The columns can be used to differentiate between greater dilutions of antibodies, like a scale from low to high going from left to right (see Figures 3 and 4 for an example). The rows of the plate can be used to test different influenza viruses against the same set of antibodies. These wells are arranged according to rows and columns (which are identified on the microtiter plate by letters and numbers, respectively). The plate contains wells (i.e., cup-like depressions that can hold a small amount of liquid) where the solution of antibodies, influenza virus and red blood cells are inserted and allowed to interact. 
(See Image 1.)Ī microtiter plate is used to perform the HI test. The HI test involves three main components: antibodies, influenza virus, and red blood cells that are mixed together in the wells (i.e., cups) of a microtiter plate. 
Scientists also use the HI test to compare the antigenic properties (i.e., the virus’ ability to be recognized by antibodies) of currently circulating influenza viruses with those of influenza viruses that have circulated in the past. HI test results can tell us whether antibodies developed after vaccination with one virus recognize, bind to, and therefore, are similar to other circulating influenza viruses. 
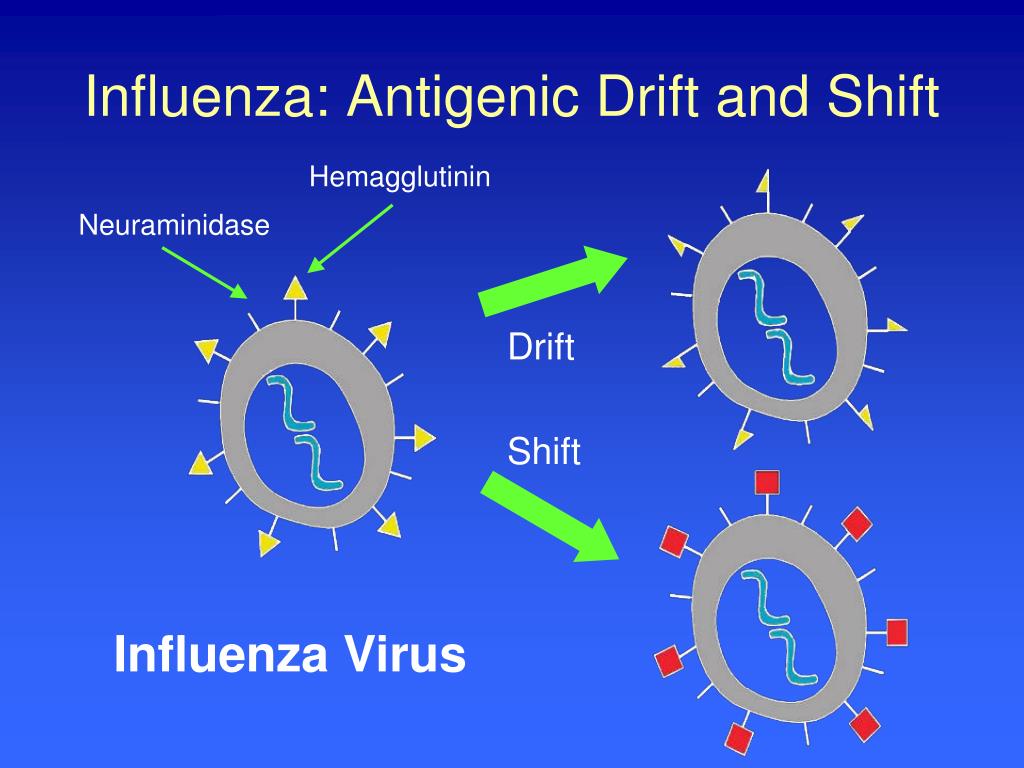
This test helps to select the candidate vaccine viruses (CVVs) included in seasonal flu vaccines. Scientists use the HI test to assess the antigenic similarity between influenza viruses. The rightmost section of the image shows what hemagglutination inhibition looks like in the well of a microtiter plate (this is explained in greater detail further below). The picture below provides an example of hemagglutination inhibition. The HI test works by measuring how well antibodies bind to the HA proteins and prevent them from “gluing” red blood cells together (i.e., hemagglutination inhibition). The rightmost section of the image shows what hemagglutination looks like in the well of a microtiter plate (this is explained in greater detail further below). The picture below provides an example of hemagglutination. HA proteins on the surface of influenza viruses can bind to red blood cells and “glue” them together, forming a lattice structure (this is known as “hemagglutination”). Scientists use a test called the hemagglutination inhibition assay (HI test) to antigenically characterize influenza viruses. The Hemagglutination Inhibition Assay (HI Test) Serology tests using human sera and genetic sequencing provide additional information about how similar circulating flu viruses are to vaccine viruses or other influenza viruses. This information also helps experts decide what viruses should be included in the upcoming season’s influenza vaccine. Antigenic characterization can give an indication of the flu vaccine’s ability to produce an immune response against the influenza viruses circulating in people. “Antigenic characterization” refers to the analysis of a virus’ antigenic properties to help assess how related it is to another virus.ĬDC antigenically characterizes about 2,000 influenza viruses during a typical flu season to monitor for changes in circulating viruses and to compare how similar these viruses are to those included in vaccines. The term “antigenic properties” is used to describe the antibody or immune response triggered by the antigens on a particular virus. When someone is exposed to an influenza virus (either through infection or vaccination) their immune system makes specific antibodies against the antigens (surface proteins) on that particular influenza virus. Two proteins (hemagglutinin and neuraminidase) on the surface of influenza viruses contain the major antigens targeted by antibodies (see Figure 1). “Antigens” are molecular structures on the surface of viruses that are recognized by the immune system and are capable of triggering one kind of immune response known as antibody production. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
